Organic Chemistry Text Book (CHEM 3401 and 3402)
- Home
-
Chapter 1: A Review of General Chemistry
Toggle Dropdown
- 1.1 Introduction to Organic Chemistry
- 1.2 Electrons, Bonds, and Lewis Structures
- 1.3 Identifying Formal Charges
- 1.4 Atomic Orbitals
- 1.5 Valence Bond Theory
- 1.6 Molecular Orbital Theory/Hybridization
- 1.7 VSEPR Theory: Predicting Geometry
- 1.8 Dipole Moments and Molecular Polarity
- 1.9 Intermolecular Forces and Physical Properties
- Problem Set
- Videos for chapter 1
- Chapter 2: Molecular Representations Toggle Dropdown
- Chapter 3: Acids and Bases Toggle Dropdown
- Chapter 4: Alkanes and Cycloalkanes Toggle Dropdown
- Chapter 5: Stereochemistry Toggle Dropdown
- Chapter 6: Chemical Reactivity and Mechanisms
- Chapter 7: Substitution Reactions Toggle Dropdown
-
Chapter 8: Addition Reactions of Alkenes
Toggle Dropdown
- 8.1 Introduction of Addition Reactions
- 8.1 Nomenclature of Alkenes
- 8.2 Hydrohalogenation of Alkenes
- 8.3 Hydration, Hydroboration, and Oxymercuration of Alkenes
- 8.4 Hydrogenation of Alkenes
- 8.5 Halogenation of Alkenes
- 8.6 Dihydroxylation, Epoxidation, and Ozonolysis of Alkenes
- Problem Set
- Chapter 8 Videos
- Chapter 9: Alkynes Toggle Dropdown
- Chapter 10: Radicals Toggle Dropdown
- Chapter 11: Synthesis Toggle Dropdown
- Problem Sets Organic Chemistry I (CHEM 3401)
-
Chapter 12: Alcohols and Phenols
Toggle Dropdown
- 12.1 Alcohol Structure
- 12.2 Solubility
- 12.3 Boiling Point & Melting Point
- 12.4 Nomenclature
- 12.5 Alcohol Acidity
- 12.6 Reactions of Alcohols and Phenols
- 12.6.1 Substitution of the Hydroxyl Hydrogen
- 12.6.2 Nucleophilic Substitution of the Hydroxyl Group
- 12.6.3 Elimination Reactions of Alcohols
- 12.6.4 Oxidation Reactions of Alcohols
- 12.6.5 Reactions of Phenols
- 12.7 Practice Problems
- 12.7.1 Alcohol Nomenclature 1
- 12.7.2 Alcohol Nomenclature 2
- 12.7.3 Alcohol Nomenclature 3
- 12.7.4 Formation of Carbonyl Compounds
- 12.7.5 Functional Relationships of Alcohols
- 12.7.6 Reactions of Alcohols & Phenols
- 12.7.7 Alcohol Reactions
- Chapter 13: Ethers and Epoxides Toggle Dropdown
-
Chapter 14: Infrared Spectroscopy and Mass Spectrometry
Toggle Dropdown
- 14.1 Introduction fo Molecular Spectroscopy
- 14.2 Infrared Spectroscopy
- 14.2.1 Introduction
- 14.2.2 Vibrational Spectroscopy
- 14.2.3 Group Frequencies
- 14.2.4 Table of Characteristic IR Frequencies
- 14.3 Mass Spectrometry
- 14.3.1 The Mass Spectrometer
- 14.3.2 Characteristics of Mass Spectra
- 14.3.3 Isotopes
- 14.3.4 Fragmentation Patterns
- 14.3.5 High Resolution Spectra
- 14.3.6 MS Practice Problems
- 14.3.6a Problem 1
- 14.3.6b Problem 2
- 14.3.6c Problem 3
- 14.3.6d Problem 4
- 14.3.6e Problem 5
- 14.3.6f Problem 6
- 14.3.6g Problem 7
- 14.3.6h Problem 8
-
Chapter 15: Nuclear Magnetic Resonance Spectroscopy and UV-Visible Spectroscopy
Toggle Dropdown
- 15.1 Nuclear Magnetic Resonance Spectroscopy
- 15.1.1 Background
- 15.1.2 Proton NMR Spectroscopy
- 15.1.2a Introduction to Proton NMR Spectroscopy
- 15.1.2b Chemical Shift
- 15.1.2c Signal Strength
- 15.1.2d Hydroxyl Proton Exchange and the Influence of Hydrogen Bonding
- 15.1.2e Pi-Electron Functions
- 15.1.2f Solvent Effects
- 15.1.2g Spin-Spin Interactions
- 15.1.2h Examples
- 15.1.3 Carbon NMR Spectroscopy
- 15.1.4 NMR Practice Problems
- 15.1.4a Problem 1
- 15.1.4b Problem 2
- 15.1.4c Problem 3
- 15.1.4d Problem 4
- 15.1.4e Problem 5
- 15.1.4f Problem 6
- 15.1.4g Problem 7
- 15.1.4h Problem 8
- 15.1.4i Problem 9
- 15.1.4j Problem 10
- 15.1.5 Table of Proton NMR Shifts
- 15.1.6 Table of Carbon NMR Shifts
- 15.2 UV-Visible Spectroscopy
- 15.2.1 Background
- 15.2.2 The Electromagnetic Spectrum
- 15.2.3 UV-Visible Absorption Spectra
- 15.2.4 The Importance of Conjugation
- 15.3 Spectroscopy Practice Problems
- Chapter 16: Conjugated Pi Systems and Pericyclic Reactions Toggle Dropdown
-
Chapter 17: Aromatic Compounds
Toggle Dropdown
- 17.1 Aromaticity
- 17.1.1 Benzene
- 17.1.2 Fused Ring Compounds
- 17.1.3 Other Aromatic Compounds
- 17.1.4 Antiaromaticity
- 17.1.5 Practice Problems
- 17.1.5a Problem 1
- 17.1.5b Problem 2
- 17.2 Reactions of Substituent Groups
- 17.2.1 Oxidation of Alkyl Side-Chains
- 17.2.2 Bromination of Alkyl Side-Chains
- 17.2.3 Reduction of Nitro Groups
- Chapter 17 Videos
-
Chapter 18: Aromatic Substitution Reactions
Toggle Dropdown
- 18.1 Electrophilic Aromatic Substitution Reactions
- 18.2 Electrophilic Aromatic Substitution Mechanism
- 18.3 Electrophilic Aromatic Substitution Activation/Deactivation and Orientation
- 18.4 Electrophilic Substitution of Disubstituted Benzene Rings
- 18.5 Practice Problems
- 18.5.1 Problem 1
- 18.5.2 Problem 2
- 18.5.3 Problem 3
- 18.5.4 Problem 4
- 18.5.5 Problem 5
- 18.5.6 Problem 6
- 18.5.7 Problem 7
- Chapter 18 Videos
-
Chapter 19: Aldehydes and Ketones
Toggle Dropdown
- 19.1 Nomenclature
- 19.2 Preparation of Aldehydes and Ketones
- 19.3 Properties of Aldehydes and Ketones
- 19.4 Reactions of Aldehydes and Ketones
- 19.4.1 Addition Reactions
- 19.4.1a Hydration
- 19.4.1b Acetal Formation
- 19.4.1c Imine Formation
- 19.4.1d Cyanohydrin Formation
- 19.4.1e Hydride Reduction
- 19.4.1f Addition of Organometallic Reagents
- 19.4.2 Reduction of Aldehydes and Ketones
- 19.4.2a Wolff-Kishner Reduction
- 19.4.2b Clemmensen Reduction
- 19.4.3 Oxidation of Aldehydes and Ketones
- 19.5 Practice Problems
- 19.5.1 Problem 1
- 19.5.2 Problem 2
- 19.5.3 Problem 3
- 19.5.4 Problem 4
- 19.5.5 Problem 5
- 19.5.6 Problem 6
- 19.5.7 Problem 7
- 19.5.8 Problem 8
- 19.5.9 Problem 9
- 19.5.10 Problem 10
- 19.5.11 Problem 11
- 19.5.12 Problem 12
-
Chapter 20: Carboxylic Acids and Their Derivatives
Toggle Dropdown
- 20.1 Nomenclature
- 20.2 Physical Properties
- 20.3 Acidity
- 20.4 Preparation of Carboxylic Acids
- 20.5 Reactions of Carboxylic Acids
- 20.5.1 Salt Formation
- 20.5.2 Substitution of the Hydroxyl Hydrogen
- 20.5.3 Substitution of the Hydroxyl Group
- 20.5.4 Reduction
- 20.5.5 Oxidation
- 20.6 Practice Problems-Carboxylic Acids
- 20.6.1 Nomenclature Practice-1
- 20.6.2 Nomenclature Practice-2
- 20.6.3 Acidity
- 20.6.4 Reactions of Carboxylic Acids
- 20.7 Carboxylic Acid Derivatives
- 20.7.1 Related Derivatives
- 20.7.2 Nomenclature
- 20.7.3 Reactions
- 20.7.3a Acyl Substitution
- 20.7.3b Nitrile Hydrolysis
- 20.7.3c Reductions
- 20.7.3d Reactions with Organometallic Reagents
- 20.7.3e Dehydration of Amides
- 20.7.4 Practice Problems-Carboxylic Acid Derivatives
- 20.7.4a Nomenclature Practice-1
- 20.7.4b Nomenclature Practice-2
- 20.7.4c Carbonyl Compounds
- 20.8 Practice Problems
- 20.8.1 Problem 1
- 20.8.2 Problem 2
- 20.8.3 Problem 3
- 20.8.4 Problem 4
- 20.8.5 Problem 5
- 20.8.6 Problem 6
-
Chapter 21: Alpha Carbon Chemistry: Enols and Enolates
Toggle Dropdown
- 21.1 Reactions at the Alpha Carbon
- 21.2 Alpha Halogenation of Enols and Enolates
- 21.3 Aldol Reaction
- 21.4 Claisen Condensation
- 21.5 Alkylation at the Alpha Position
- 21.5.1 Enolate Alkylation
- 21.5.2 Dicarbonyl Alkylation
- 21.5.3 Decarboxylation Following Alkylation
- 21.5.4 Conjugate Reactions
- 21.5.4a Michael Reaction
- 21.5.4b Robinson Annulation
- 21.5.4c With Hydrides and Organometallics
- 21.6 Practice Problem
- 21.6.1 Problem 1
- Org Chem II - Problem Sets - Collection (CHEM 3402)
- Problem Set
6.5 Carbocation Rearrangements
In the first half of the nineteenth century it was generally believed that reactions of organic compounds proceeded with minimal structural change. This tenet simplified the elucidation of the numerous substitution, addition and elimination reactions that characterized the behavior of common functional groups. However, subsequent discoveries showed that nature was not always so obliging, leaving chemists and chemistry students to grapple with the possibility of deep seated structural change occurring during certain reactions. A large number of these structural rearrangements are triggered by intermediates incorporating positively charged or electron deficient atoms, which in the case of carbon are carbocations. Two such examples, already noted, are the addition of HCl to 3,3-dimethyl-1-butene and forced hydrolysis of neopentyl bromide. This chapter will describe and discuss other cases of this intriguing group of transformations.
1. Wagner-Meerwein Rearrangements
The chemical behavior of neopentyl bromide, 2,2-dimethyl-1-bromopropane, is an instructive place to begin this discussion. The very low SN2 reactivity of this 1º-bromide was noted earlier, and explained by steric hindrance to the required 180º alignment of reacting orbitals. Under conditions that favor SN1 reactivity, such as solution in wet formic acid, neopentyl bromide reacts at roughly the same rate as ethyl bromide. Both of these compounds are 1º-alkyl halides, and for an SN1 reaction the rate determining step requires ionization to a 1º-carbocation. As noted in the carbocation stability order shown below, such carbocations are relatively unstable and are formed slowly. The product from ethyl bromide is ethanol, the simple and direct substitution product, but neopentyl bromide yields 2-methyl-2-butanol instead of the expected neopentyl alcohol. A change in the way the five carbon atoms in this product are bonded to each other has clearly taken place.
|
Once formed, the ethyl cation can only be transformed by a substitution or elimination process. In the case of the neopentyl cation, however, the initially formed 1º-carbocation may be converted to a more stable 3º-carbocation by the 1,2-shift of an adjacent methyl group with its bonding electrons. A mechanism demonstrating such a rearrangement is shown below, and it explains the overall structural changes very nicely.

Increasing the stability of carbocation intermediates is not the only factor that leads to molecular rearrangement. If angle strain , torsional strain or steric crowding in the reactant structure may is relieved by an alkyl or aryl shift to a carbocation site, such a rearrangement is commonly observed. The following examples illustrate rearrangements induced by the strain in a small ring. Although a 3º-carbocation is initially formed, the angle and torsional strain of the four-membered ring is reduced by a methylene group shift resulting in ring expansion to a 2º-carbocation. Clicking on the equation diagram will display a mechanism for these transformations.

Following the ring expansion step other reactions may take place, depending on the conditions. In aqueous acid the rearranged 2º-carbocation may bond to a water nucleophile, producing a 2º-alcohol, lose a proton to water, giving 3,3-dimethylcyclopentene (not shown), or undergo a second rearrangement to a 3º-carbocation, which then forms 1,2-dimethylcyclopentene. Indeed, it is not uncommon to encounter sequences of rearrangements in more complex compounds, and these may produce products with structures remarkably different from that of the starting compound. The following equation shows one such reaction. A curved arrow representation of the five sequential ring expansion steps will be added to the equation by clicking on the diagram.

In the terminology of pericyclic reactions, 1,2-alkyl shifts of this kind are classified as [1,2]-sigmatropic shifts. Since this is a two-electron process (the 2 electrons in the relocated sigma bond), the rearrangement is predicted to be suprafacial. Considerable evidence supporting this conclusion has been obtained, as the following example shows. Protonation of the double bond gives a 3º-carbocation. An adjacent hydrogen atom (colored blue) shifts as a hydride moiety to create a new 3º-carbocation, which in turn induces the shift of a methyl group (colored green) with formation of yet another 3º-carbocation. This electrophilic center then bonds to the nucleophilic oxygen of the carboxylic acid function, releasing a catalytic proton to continue the process. Because of the fused polycyclic structure of this compound, the relative orientation of the migrating groups is easily determined, and is seen to be suprafacial. Rearrangements consisting of consecutive 1,2-shifts often take place in a concerted, and therefore stereospecific fashion; however, it must not be assumed that the group shifts are simultaneous. Each shift involves a separate transition state in which the positive charge is delocalized over the migration terminus, origin and migrating group.

Many of the most interesting rearrangements of this kind were discovered during structural studies of naturally occurring compounds. Among these the terpenes presented numerous remarkable reactions, and the names of two chemists who were instrumental in unraveling their complex transformations, H. Meerwein and G. Wagner, are permanently associated with these rearrangements. The addition of gaseous HCl to α-pinene proved particularly puzzling to these early chemists. Under ordinary conditions, this liquid component of turpentine gave a crystalline C10H17Cl compound, originally called "artificial camphor", now known as bornyl chloride. An unstable isomer, pinene hydrochloride, can be isolated under mild conditions, but it rapidly isomerizes to bornyl chloride. Treatment of bornyl chloride with base gave a crystalline isomer of pinene called camphene, together with small amounts of another unsaturated hydrocarbon (bornylene). Addition of HCl to camphene, in a similar fashion, initially produces an unstable chloride (camphene hydrochloride) which quickly isomerizes to isobornyl chloride, a stereoisomer of bornyl chloride. We now know that bornyl chloride and isobornyl chloride are endo / exo-2-chloro isomers of the 1,7,7-trimethylbicyclo[2.2.1]bicycloheptane system. Structural formulas for these compounds are drawn below, along with camphene, the rearranged elimination product.

Mechanisms for these rearrangements will be pictured by clicking on the above diagram. In the new display we see that both pinene and camphene form 3º-carbocations when the double bond is protonated. Rearrangement to a 2º-carbocation is favored by relief of small-ring strain in the case of pinene, and relief of steric congestion in the case of camphene. However, this is an oversimplification which ignores the fact that these reactions take place in nonpolar solvents, and are unlikely to involve discrete, unassociated carbocations. Some of the stereoelectronic effects that influence these reactions will be shown by clicking on the above diagram a second time. Structures for the initially formed unstable hydrochlorides of pinene and camphene are drawn on the left. Optimal orbital overlap of breaking and forming bonds requires rear-side approach of the shifting alkyl group to the site of the leaving chloride anion, in a manner similar to a SN2 reaction. The chloride anion is located on one side of the carbocation formed by the alkyl shift, and immediately bonds to that face of the tricoordinate carbon. In this view of these rearrangements, the chloride anion never escapes the attractive influence of its cationic partner, and the product stereoselectivity is understandable. Lewis acid catalysts (e.g. FeCl3) catalyze these rearrangements, and the product favored at equilibrium is bornyl chloride.
The rearrangement that occurs under base catalyzed elimination conditions reflects the eclipsed configuration of the two-carbon bridge bearing the chlorine atom. Because of this configuration, the anti-coplanar structure favored by the E2 transition state cannot be achieved. Syn elimination gives a small amount of bornylene, but rearrangement to a camphene precursor predominates. Repeated clicking on the above diagram will cycle the displays.
Yet another example of the remarkable acid-catalyzed rearrangements found to occur with terpenes was observed in a study of the sesquiterpene caryophyllene (from oil of cloves). Here it is evident that reactive sites may interact and form bonds from one side of a medium-sized ring to another side. The mechanisms for many such rearrangements have been, and still are studied with great interest.

2. Pinacol Rearrangement
The pinacol rearrangement was the first molecular rearrangement identified as such by early chemists. The defining example of a pinacol rearrangement is shown in the following diagram. Pinacol itself is produced by magnesium reduction of acetone, probably by way of a ketyl intermediate. Since the diol is symmetrical, protonation and loss of water takes place with equal probability at either hydroxyl group. The resulting 3º-carbocation is relatively stable, and has been shown to return to pinacol by reaction in the presence of isotopically labeled water. A 1,2-methyl shift generates an even more stable carbocation in which the charge is delocalized by heteroatom resonance. Indeed, this new cation is simply the conjugate acid of the ketone pinacolone, which is the product of repeated rearrangements catalyzed by proton transfer. Each step in this rearrangement is potentially reversible, as demonstrated by the acid catalyzed dehydration of pinacolone (and pinacol) to 2,3-dimethyl-1,3-butadiene under vigorous conditions.
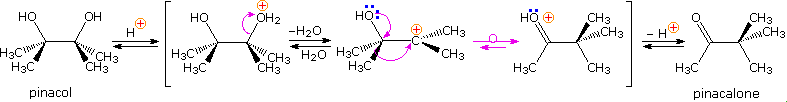
Many factors must be considered when analyzing the course of a pinacol rearrangement. These include:
• Which hydroxyl group is lost as water? or Which intermediate carbocation is more stable?
• What is the inherent shifting tendency (migratory aptitude) of different substituent groups?
• What is the influence of steric hindrance and other strain factors on the rearrangement?
• Are epoxides formed as intermediates in the pinacol rearrangement?
• Does product stability govern the outcome of competing rearrangements?
• Do the reaction conditions (i.e. type of acid, concentration, solvent and temperature) influence the course of rearrangement?
Virtually all of these factors have been shown to be important in one or more cases, and a full analysis of their complex interaction is beyond the scope of this text. Nevertheless, a few examples will be presented to demonstrate the general nature of this transformation, and to illustrate the action of some of the above factors. In the first reaction shown below, we see an example of kinetic versus thermodynamic product control. Under mild acid treatment, the diol rearranges rapidly to an aldehyde by way of a 1,2-hydrogen shift to the initially formed diphenyl 3º-carbocation. More vigorous acid treatment of the diol or the aldehyde generates the more stable phenyl ketone (conjugation of the phenyl and carbonyl groups). Mechanisms for this and the other reactions will be presented by clicking on the diagram. A pink colored arrow designates rearrangement; light blue arrows indicate epoxide ring closing or opening reactions. Repeated clicking toggles the reaction and mechanism displays.
The second example describes a similar reacting system, which provides additional information from stereochemical and isotopic labeling features. Loss of water from the 3º-carbinol site, followed by a reversible 1,2-hydride shift, generates the conjugate acid of the ketone product. At short reaction times, racemization of recovered diol starting material occurs at the same rate as rearrangement. A corresponding phenyl shift to the initially formed 3º-carbocation generates the aldehyde conjugate acid, and the aldehyde itself has been shown to isomerize to the same rearranged ketone under the conditions of this pinacol rearrangement. An isotopic carbon label (colored green) in either the diol or aldehyde is scrambled (colored brown) in the course of these reactions, suggesting an epoxide intermediate.
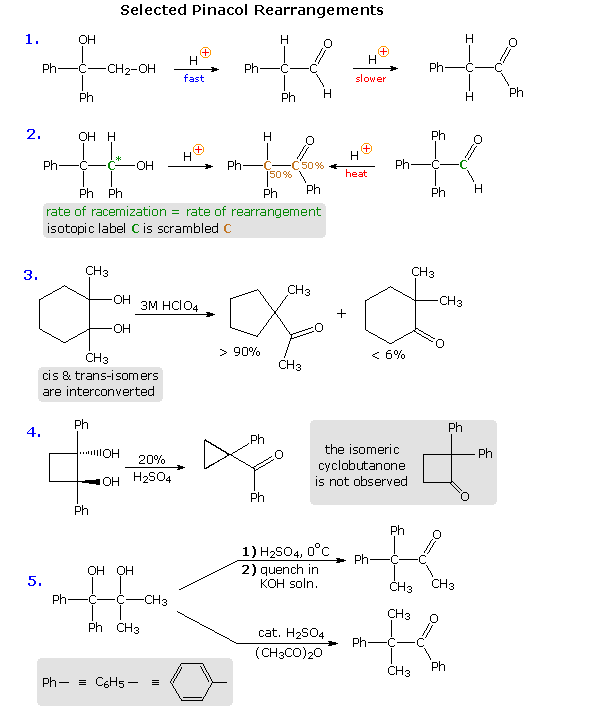
In reaction # 3 either the cis or trans diol may be used as a reactant. These isomers are rapidly interconverted under the rearrangement conditions, indicating that the initial water loss is reversible; a result confirmed by isotopic oxygen exchange. The clear preference for a methylene group shift versus a methyl group shift may reflect inherent migratory aptitudes, or possibly group configurations in the 3º-carbocation intermediate. In the conformation shown here both methyl and methylene groups may shift, or an epoxide ring may be formed reversibly. An alternative chair-like conformation having an equatorial methyl group should be more stable, but would not be suitable for a methyl shift. The predominant ring contraction is therefore understandable. Reaction # 4 is an unusual case in which a strained ring contracts to an even smaller ring. Phenyl groups generally have a high migratory aptitude, so the failure to obtain 2,2-diphenylcyclobutanone as a product might seem surprising. However, the carbocation resulting from a phenyl shift would be just as strained as its precursor; whereas the shift of a ring methylene group generates an unstrained cation stabilized by phenyl and oxygen substituents. Conjugative stabilization of the phenyl ketone and absence of sp2 hybridized carbon atoms in the small ring may also contribute to the stability of the observed product.
Finally, reaction # 5 clearly shows the influence of reaction conditions on product composition, but explaining the manner in which different conditions perturb the outcome is challenging. Treatment with cold sulfuric acid should produce the more stable diphenyl 3º-carbocation, and a methyl group shift would then lead to the observed product. The action of a Lewis acid in acetic anhydride, on the other hand, may selectively acetylate the less hindered dimethyl carbinol. In this event the acetate becomes the favored leaving group (presumably coordinated with acid), followed by a 1,2-phenyl shift. It is reported that the symmetrically substituted isomeric diol (drawn in the shaded box) rearranges exclusively by a methyl shift, but the configuration of the starting material was not stated (two diastereomers are possible).
Because of the influence of other factors (above), it has not been possible to determine an unambiguous migratory order for substituents in the pinacol rearrangement. However, some general trends are discernible. Benzopinacol, (C6H5)2C(OH)C(OH)(C6H5)2, undergoes rapid rearrangement to (C6H5)3CCOC6H5 under much milder conditions than required for pinacol. Indeed, it is often the case that phenyl or other aromatic substituents adjacent to a forming carbocation will facilitate that ionization in the course of their migration to the cationic site. In non-aromatic compounds of the type (CH3)2C(OH)C(OH)RCH3, migration of R increases in the manner R = CH3 < R = C2H5 << R = (CH3)3C. Since the shifting alkyl group must carry part of the overall positive charge, alkyl substitution should have a stabilizing influence on the rearrangement transition state. Finally, fluorine substitution, as in C6H5(CF3)C(OH)C(OH)(CF3)C6H5 renders the diol unreactive under acid catalyzed rearrangement conditions. Here, the powerful inductive withdrawal of electrons by fluorine inhibits positive charge formation.
